





|
|
|
|
April 30, 2007
CFLs are for screwing, while teeth are for chewing
The amount of mercury in the mouth of a person with fillings was on average 2.5 grams, enough to contaminate 5 ten acre lakes to the extent there would be dangerous levels in fish.I really like Steven Milloy. But I also like my CFLs. And while I don't want the latter to be made mandatory, I don't find Milloy's mercury satire (if indeed it is that) to be at all persuasive one way or the other on CFLs. Especially in view of my mouth: 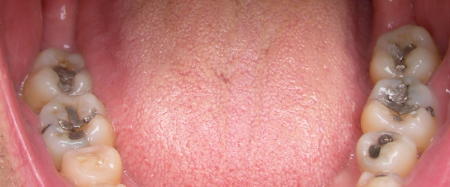 Will someone please tell Steve Milloy that your average American mouth is potentially far, far more hazardous than an ordinary CFL? I see that Glenn Reynolds has pointed out the problem with Milloy's argument -- time and time again -- but I offer real, tangible proof, not just facts and figures. Compare the mercury in my mouth to the actual CFL mercury content Glenn cited: "The very small amount of mercury in a CFL -- about 5 milligrams, compared to an old-fashioned home thermometer, which had about 500 milligrams -- is safe while the bulb is in operation and poses little risk even if it breaks, according to the U.S. Environmental Protection Agency."Glenn follows up with this: I'd be interested in seeing more on this topic, but if CFLs were as deadly as Milloy suggests, I wouldn't expect big companies to sell them for fear that the trial lawyers would take them to the cleaners. I kind of think that Milloy is just having a bit of fun turning enviro-scare tactics back upon themselves, but I don't think there's much foundation to these worries.I agree, and I like to turn around enviro-scare tactics back upon themselves too. Which is why I don't think Milloy went far enough. (And why I'll continue to use both my CFLs and my teeth!) Seriously, I don't mean to get hysterical about this, but the fact is, my teeth have 500 times the amount of mercury as does a CFL! Add to that the fact that I don't chew my CFLs. I screw them in and they just sit there emitting light inexpensively, and they never burn out. Far from proving that CFLs are actually dangerous, Milloy's remarks give a good idea of how hysterical the mercury bureaucracy has become. (Not a new issue for me.) Sheesh. Could someone please tell me why we're not all dead? (I'm tempted to say that they can have my CFLs, AND my teeth when they pull them from their respective sockets, but as I'm already up to my neck defending my guns and Coco's ovaries, I don't want to be defending myself against more threats than my corpse can handle.) UPDATE: Readers who like CFLs, be sure to join Pajamas Media's One Billion Bulbs campaign.  posted by Eric on 04.30.07 at 02:47 PM
Comments
I remember the day I learned that bacon and Scotch were accused of being carcinogens. I decided to stop worrying. To Hell with it. That was a couple of decades ago and I'm still here. Enjoy! [crunch, slurp]. Bleepless · April 30, 2007 10:20 PM decay tooth http://www.url.chefhost.com/87c7ae > tooth decay tooth decay · May 29, 2007 04:15 PM Post a comment
You may use basic HTML for formatting.
|
|
May 2007
WORLD-WIDE CALENDAR
Search the Site
E-mail
Classics To Go
Archives
May 2007
April 2007 March 2007 February 2007 January 2007 December 2006 November 2006 October 2006 September 2006 August 2006 July 2006 June 2006 May 2006 April 2006 March 2006 February 2006 January 2006 December 2005 November 2005 October 2005 September 2005 August 2005 July 2005 June 2005 May 2005 April 2005 March 2005 February 2005 January 2005 December 2004 November 2004 October 2004 September 2004 August 2004 July 2004 June 2004 May 2004 April 2004 March 2004 February 2004 January 2004 December 2003 November 2003 October 2003 September 2003 August 2003 July 2003 June 2003 May 2003 May 2002 AB 1634 See more archives here Old (Blogspot) archives
Recent Entries
• Lubos Motl Looks At Sun Spots
• IEC Fusion for Dummies • Harmonica Joe • The Last Full Measure • CO2 - Its In The Air • Staged? • Latest Fusion News • The Future Of Mainstream Media: 1968 • Clouds In Chambers • More Uncertain
Links
Site Credits
|
|
I don't think I trust their math, so let's doublecheck it.
The FDA limit for mercury in fish is 1 part-per-million (and that limit is much lower than the level required to cause actual illness).
5 ten acre lakes (which is itself a problem, since an "acre" is not a measure of volume, and a lake covering ten acres is of unstated depth and thus volume; in other words, there's no way to just guess how much water these five lakes contain!)?
If we assume an average depth of 3 feet (shallow?) we end up with about 37,000 cubic meters of water per lake, thus about 185,000 cubic meters in the five lakes.
Thus, 185 million liters of water. For 1 ppm, that's one milligram per liter, which requires 185 kilograms of mercury, to get 1 ppm in the water - but the fish will of course concentrate it.
Now, the question is, which fish get how much mercury from how much water?
Evidently, "According to the EPA, fish at the top of the aquatic food chain bioaccumulate methylmercury to a level approximately 1 million to 10 million times greater than dissolved concentrations found in surrounding waters."
So we divide that 185,000 grams by a million, to get .185 grams (of dissolved methylmercury).
Of course, the problem with that is that the mercury in your mouth is not aqueous methyl-mercury, and is not accessible the way dissolved mercury is.
Much like saying that the lead in one bullet is sufficient to contaminate a small reservoir with lead... it's true at the mathematical level, but false in application, because amalgam, just like lead, isn't soluble.
(And because you're not a fish at the top of the food chain, and out-gassed mercury from amalgam fillings is more likely to be exhaled than inhaled unless you're a mouth-breather...)